Fantastic paper from Brooks Able at Berkeley using an organic catalyst to achieve high molecular weight for poly(THF) via cationic ring-opening polymerization. The low ceiling temperatures of these polymers make them attractive targets for reversible polymerizations, but it has previously been really hard to get to the molecular weights required for good mechanical properties. #chemistry #polymers #JACS https://pubs.acs.org/doi/10.1021/jacs.5c18359
#JACS
A novel bifunctional molecule induces selective nitrosation of BTK. Mechanistically, it first covalent bind to BTK and the release NO, which yielded in the nitrosation of a cysteine side chain. Published in #JACS by Jiangbing Wu, Zhangjian Huang and coworkers (#China)
Read more on doi: 10.1021/jacs.5c07409 or on my page: https://me.organicchemistry.eu/post/nitrosation-jacs-2025/
When you think of a reaction between an acid and an alcohol, you think of an ester as the product. MacMillan and co-workers from Princeton have now described a different pathway: Selective decarbonylative C-C coupling via a metal-catalyzed photoredox reaction. Read more about this in JACS:
Novel enantioselective #synthesis of rubriflordilactone B by Gui and colleagues from the Chinese Academy of Science published in #JACS
This is already the second total synthesis of this group, with an interesting reaction cascade as the key reaction in this process.
Luo et al. from Tsinghua University (#China) report in #JACS a novel method to create enantioenriched α-amino aldehydes using a chiral amine catalyst and iridium photocatalysis. Through this strategy α-amino aldehydes with up to 93% ee could be synthesised and subsequently stabilised via analogues formation (e.g. through Wittig reaction).
https://me.organicchemistry.eu/post/deracemization-aldehyde-jacs-2025/
Joseph Tuccinardi and John Wood from Baylor University (#USA), introduced a novel strategy to efficiently synthesize the core scaffold of aleutianamine. Their study involved a vinylogous Mukaiyama-Michael reaction cascade using two previously unknown coupling partners. Published in #JACS
https://me.organicchemistry.eu/post/aleutianamine-jacs-2025/
Another total synthesis in the spotlight: Already early this year Mingji Dai and his research team described the total synthesis of heilonine in #JACS (doi: 10.1021/jacs.3c13492). This synthesis is short and elegant, using multiple C-H functionalisations and a Nazarov cyclisation for their key step.
Finaalllyyyy.... The first 𝐉𝐀𝐂𝐒 (IF - 14.5)😍🤩...
Special thanks to Felipe Fantuzzi (need any introduction? 3rd paper together🫂) & Holger group (University of Würzburg), Sougata Sarkar Sir (without whom I'd not even have an M.Sc.🙏)... & my mother (for insisting on asking Felipe to start a collaboration)😘😍
Link - https://pubs.acs.org/doi/10.1021/jacs.4c10967
#ACS #jacs #organometallic #computational #collaboration #journal #article #openaccess
Efficient Synthesis of ProTide antivirals: A team from from #Fudan University and #Shanghai Jiao Tong University reporting a copper-catalyzed method for synthesizing chiral phosphates in #JACS.
This research represents a new tool for the synthesis of ProTide nucleoside analogues, providing an efficient route that can enhance the development of antiviral drugs in future studies.
My evening lecture: Total synthesis of cylindrocyclophane A by Davies and Craig published in #Science and the stereoselective synthesis of protides by Shang published in #JACS
The construction of quaternary centres via C-C bond formation is a major challenge. A new stereoselective method using lithium enolates and Ru-catalysed allylic substitution has been reported in #JACS by the group of Erick Carreira (ETH #Zürich). This promising approach provides access to these sterically crowded motifs.
Terpenoids are a large class of secondary #NaturalProducts, with some possessing an interesting polycyclic core that has attracted research interest. Researchers from #Shandong #University in #China led by Ze-Jun Xu (徐泽军) and Hong-Xiang Luo (娄红祥) have now reported the #TotalSynthesis of platensilin and platensimycin, as well as the formal synthesis of platencin, using a biomimetic approach from a common precursor.
Read more in #JACS: https://pubs.acs.org/doi/10.1021/jacs.4c02256
My Reading Tip of the Week:
Scabrolide A, a polycyclic marine natural product, has fascinated chemists due to its highly constrained structure. Recently, Pengfei Hu and colleagues from Westlake University in Hangzhou, China, published an innovative radical cascade strategy for efficiently synthesizing the scaffold of this polycyclic compound.
Divergent Synthesis of Scabrolide A and Havellockate via an exo-exo-endo Radical Cascade
Happy to see this cover I made for the nice work of Raquel Galian and colleagues published in the American Chemical Society Journal #JACS! The article shows how #BODIPY dyes anchored to #perovskite nanocrystals behave differently depending on their structure, showing #HotCarriers extraction and Förster resonance energy transfer (#FRET).
✏ here the link to the paper: https://pubs.acs.org/doi/10.1021/jacs.3c14335
Synthesis of Polysubstituted Bicyclo[1.1.0]butanes by Ilan Marek (#Israel Institute of Technology) published in #JACS
Der Krankenhauskeim 𝘗𝘴𝘦𝘶𝘥𝘰𝘮𝘰𝘯𝘢𝘴 𝘢𝘦𝘳𝘶𝘨𝘪𝘯𝘰𝘴𝘢 🦠 ist auch wegen seiner Resistenz gegen zahlreiche #Antibiotika gefährlich. Ein Forschungsteam der @HHU und des Forschungszentrums Jülich hat nun einen Mechanismus gefunden, der es ermöglicht, die Virulenz des Keims abzuschwächen. Auf diesem Wissen aufbauend kann ein neuer Ansatz für Antibiotika 💊 entwickelt werden, wie die Autoren in der Fachzeitschrift #JACS Au darlegen. https://www.fz-juelich.de/de/aktuelles/news/meldungen/2024/neuer-ansatz-fuer-antibiotikaentwicklung
Melognine was thought to be a highly complex alkaloid with a polycyclic ring system. Yui Irie and Satoshi Yokoshima (Nagoya University, #Japan) published a total synthesis of the proposed structure in the journal #JACS. However, the synthesised structure turned out to have different analytical properties. The authors concluded that melognine is actually identical to melodinine L. Check out the intriguing total synthesis route on JACS:
https://pubs.acs.org/doi/10.1021/jacs.4c02086
If you have some time, take a look at the total synthesis of Hunterine A by Elliot Hicks, Kengo Inoue and Brian Stoltz published in #JACS (#OpenAccess). Very elegant synthesis and late-stage cyclization strategy.
⚗️ Using the Bunsen burner for the final step of a complex total synthesis?
📰 #OpenAccess #TotalSynthesis of Euphorikanin A by Erick Carreira and colleagues from #ETH Zurich (#Switzerland) published in #JACS. A late-stage atrospecific cascade reaction led to the core structure of the terpene and pyrrolysis finally yielded the natural product.
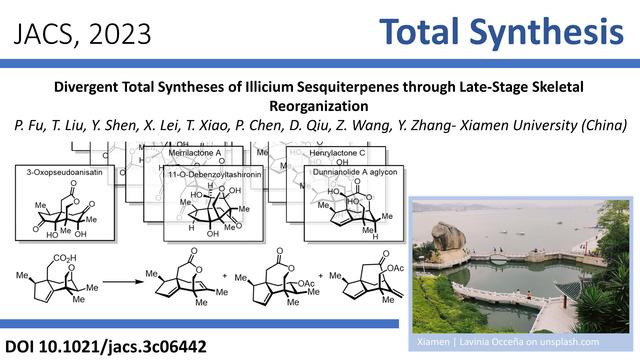
The #TotalSynthesis of highly condensed polycyclic terpenoids has been a hot topic in #Chemistry recently. Yandong Zhang (#Xiamen University, #China) and co-workers now were able to synthesize such complex terpenes by epoxide-ene cyclization, oxygenation, and subsequent skeletal rearrangement. Read more about this protecting group free total synthesis in #JACS: https://pubs.acs.org/doi/10.1021/jacs.3c06442



![The presence of inherent electronic unsaturation in aluminum predominantly results in the formation of aluminum clusters, with very few examples of compounds containing discrete chains of aluminum atoms in existence. In this work, we present the successful synthesis and structural authentication of a highly unusual trialane species with a near-linear chain of three Al atoms, alongside a carbene-stabilized aluminyl anion ([LAlR2]−), an alternative product produced by varying the reaction conditions. Quantum-chemical calculations have been applied to elucidate the electronic structure and bonding of these novel compounds. Additionally, we successfully trapped a reaction intermediate using an alkyne, suggesting the intermediacy of a base-stabilized monomeric alumylene (LRAl:), which is also investigated through computational methods.](https://files.mastodon.social/media_attachments/files/113/552/118/452/095/584/small/dd7f1573a374ec8b.jpg)






![Structure of Bicyclo[1.1.0]butanes](https://files.mastodon.social/cache/media_attachments/files/112/471/720/504/408/516/small/399ed9ea2b55d174.png)